In the world of engine maintenance, oil additives are the unsung heroes. They perform critical jobs that keep your engine running smoothly and extend its life. In this article, Steven Lumley, technical manager at WearCheck, focuses on one of the most important groups of additives: detergents.
What Do Detergents Do?
We rely on detergents every day to wash our clothes and clean our homes. In a similar way, detergents in engine oil act as powerful cleansing agents. They are specialized, oil-soluble compounds that work to keep your engine’s internal parts free of harmful deposits.
Specifically, detergents work in the high-temperature areas of the engine, such as the piston rings and cylinder liners, to:
Prevent deposits: They keep surfaces clean by preventing sludge and varnish from forming.
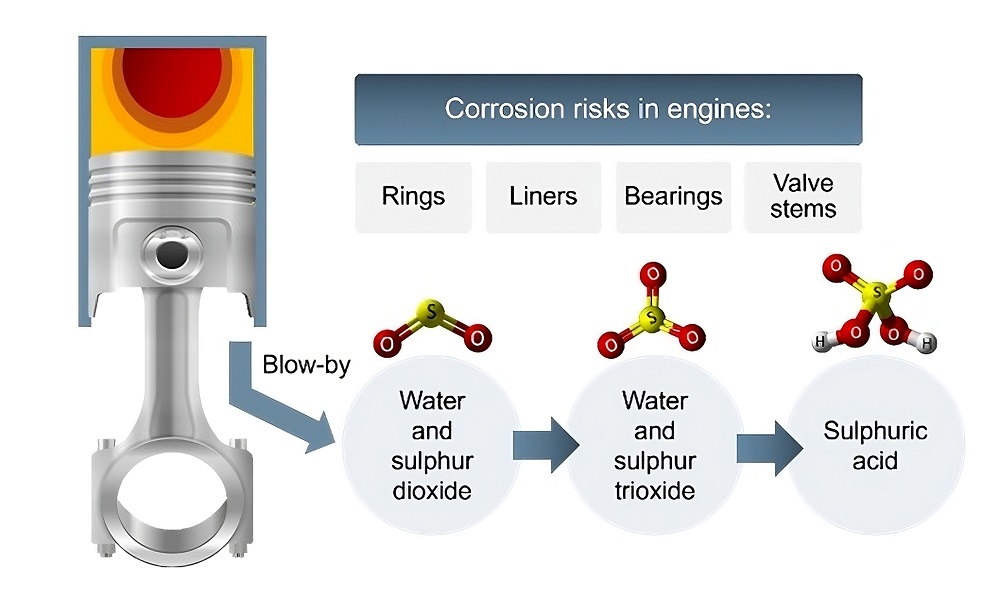
Neutralize acids: They react with and neutralize harmful acids created during the combustion process, which helps prevent corrosion.
Provide rust protection: This is a crucial function that works alongside acid neutralization to protect metal surfaces.
How Do They Work?
The cleaning action of detergents is based on their unique chemical structure. They are organo-metallic compounds, meaning they contain a metal (like calcium or magnesium) within an organic molecule.
Detergent molecules have a polar head and a long, non-polar tail. Think of the polar head as a tiny magnet that is attracted to metal surfaces and deposits, which are also polar. The detergent’s polar head has a stronger charge, allowing it to “pull” deposits away from metal surfaces and hold them in suspension within the oil.
This is a powerful partnership. Once detergents remove the deposits, they work with another type of additive, dispersants, to prevent these deposits from clumping together and settling back onto engine parts. Dispersants essentially keep the contaminants suspended and dispersed throughout the oil until they can be removed during an oil change. Together, detergents and dispersants are the “power couple” of the engine-oil additive world.
A Deeper Dive into the Chemistry
Detergents are composed of a surfactant and a colloidal inorganic phase, which gives them a micellar structure.
The basic colloidal core of the detergent is what neutralizes acids. When fuels burn, they produce harmful acids like nitric and sulfuric acid, which can cause significant wear and corrosion. The oil’s ability to neutralize these acids is measured by its Total Base Number (TBN). The higher the TBN, the greater the oil’s acid-neutralizing capacity.
The surfactant component of the detergent creates a protective layer on metal surfaces. This layer not only prevents new deposits from forming but also inhibits rust and corrosion.
The Future of Detergents: Low-SAPS Oils
Newer engine designs, especially those with exhaust after-treatment systems like Diesel Particulate Filters (DPFs), have changed the requirements for engine oils. These systems are sensitive to SAPS (Sulphated Ash, Phosphorus, and Sulphur) levels.
Because traditional detergents contain metals, they can produce ash when they burn in the engine. This ash contributes to the overall SAPS level of the oil and can eventually clog and damage DPFs. To protect these modern emission systems, the industry is moving toward low-SAPS engine oils.
This shift is leading to the development of new, alternative chemistries with a greater focus on ashless, metal-free detergents. These innovations are crucial for ensuring that engine oils can keep pace with advancements in engine and emission-control technology.

